University researchers discover pathway that regulates growth of nerve cells along spinal cord
By John EastonMedical Center Public Affairs
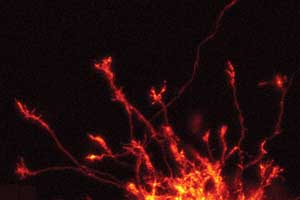 The spinal cord’s commissural neurons, which receive sensory signals such as pain from the primary neurons in other regions of the body, have a complex growth process. Growth occurs at the growth cones (pictured above) found at the tip of each axon. Receptors in the growth cones detect extracellular signals and grow toward or away from the source. |
A University research team has discovered a crucial signaling pathway that controls the growth of nascent nerves within the spinal cord, guiding them toward the brain during development.
The study, published in the Friday, Dec. 12 issue of the journal Science, solves a long-standing scientific mystery. It may also help restore function to people with paralyzing spinal cord injuries.
“This is the first guidance mechanism that regulates growth of nerve cells up and down the spinal cord,” said Yimin Zou, Assistant Professor in Neurobiology, Pharmacology & Physiology at the University.
“This is exciting to scientists because these neurons are the primary model system we use to understand assembly of the nervous system,” he said. “It’s exciting to clinicians because it could help regenerate damaged axons in the central nervous system.”
The study focused on commissural neurons, which are found in the spinal cord. These neurons receive sensory signals such as pain, heat or cold from the primary neurons that reach from the hands or feet, for example, to the spinal cord. The commissural neurons relay those signals up the spinal cord to the nerve cells that process the information in the brain.
In a meticulous series of experiments with rats, Zou and colleagues show that a gradient of chemoattractant(s) along the spinal cord, probably formed by one or multiple Wnt proteins, lures growing commissural neurons toward the brain.
The Wnt family of proteins carries signals from cell to cell, regulating the interactions between cells during many development processes. Wnt proteins bind to receptors of the “Frizzled” family on the cell surface.
In the Science paper, Zou and his colleagues show that the Wnt gradient is detected by a receptor known as Frizzled3, found at the tips of these growing neurons. Commissural axons in Frizzled3-deficient mice lost directionality of growth along the spinal cord.
If Wnt proteins could be used to entice damaged commissural neurons to regenerate and restore the connections between nerve cells of the spinal cord and the brain, it could revolutionize treatment of paralyzing spinal cord injuries.
Many researchers are studying ways to use stem cells to regenerate damaged tissues. Even if stem cells can be successfully “trained” to become the type of neurons needed and transplanted into the damaged central nervous system, “they still need to be guided precisely to their targets in order to rebuild the connections,” explained Zou. “Understanding how the brain and spinal cord are connected during embryonic development should give us clues about how to repair these connections in adulthood.”
 Yimin Zou |
But, “this is just half of the battle,” Zou cautioned. A spine-injured patient would also have to rebuild the other nerves, which carry messages from the brain to the spinal cord, such as the corticospinal tracts. The cues that steer these brain axons down the spinal cord have not yet been identified.
Scientists have long wondered how something as complex as the human nervous system, with more than 100 billion neurons, each connected to a thousand or more target cells, gets correctly assembled.
These recent findings will allow scientists to also explore “how growth cones undergo remodeling during navigation so that they constantly adjust the direction of their growth,” Zou added.
“This should help explain how the complicated connections in our nervous system are established and potentially lead to ways to remobilize the guidance programs to repair the damaged circuits in adulthood.”
![[Chronicle]](/images/sidebar_header_oct06.gif)